News
Your source for the latest updates, breakthroughs, and insights in drug development and manufacturing. Explore our featured stories, press releases, and event announcements to stay informed about our innovations and contributions to drug development and manufacturing advancements.
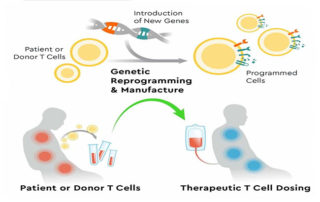
Kyverna Therapeutics Submits IND for Novel CAR T-Cell Therapy to Treat Lupus Nephritis
Company files its first Investigational New Drug application for its lead program KYV-101, a novel fully human CD19 CAR T-cell therapy, for the treatment of lupus nephritis. Kyverna’s therapeutic platform combines advanced T-cell engineering and synthetic biology technologies to suppress and eliminate...
A report on the state of the cell and gene therapy industry
Twice a year, the Alliance for Regenerative Medicine (ARM) surveys the cell and gene therapy landscape and ARM’s initiatives to advance the sector and provide an update to their members, their stakeholders, and the public at large. While external conditions ebb and flow, the sector’s progress — in clinical milestones, regulatory developments, and patient impact — steadily advances...
Kyverna Therapeutics’ $85 million series B financing reflects significant investor confidence in the science behind cell therapies
Kyverna Therapeutics, a cell therapy company engineering a new class of therapies for serious autoimmune diseases, today announced it has closed an oversubscribed $85 million Series B financing round led by Northpond Ventures. Additional investors included the company’s founding [...]
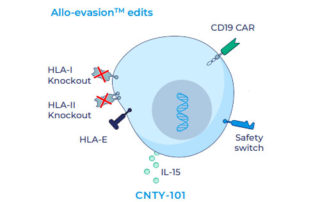
First allogeneic cell therapy candidate engineered to target cd19 for b-cell malignancies receives IND clearance, will proceed to clinical trials
Century Therapeutics, an innovative biotechnology company developing induced pluripotent stem cell (iPSC)-derived cell therapies in immuno-oncology, announced today that the company has been notified by the U.S. Food and Drug Administration (FDA) that the Company’s ELiPSE-1 clinical study may proceed [...]
FDA approves the first gene therapy for people with beta-thalassemia who require regular red blood cell transfusions
bluebird bio has announced the the U.S. Food and Drug Administration (FDA) has approved ZYNTEGLO® (betibeglogene autotemcel), also known as beti-cel, a one-time gene therapy custom-designed to treat the underlying genetic cause of beta‑thalassemia in adult and pediatric patients who [...]
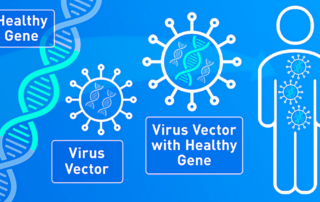
How gene therapy can cure or treat diseases
The genes in your body’s cells play a key role in your health. Indeed, a defective gene or genes can make you sick. Recognizing this, scientists have worked for decades on ways to modify genes or replace [...]